By: Dr. Ralph Lazarus R. Rapacon, FPCS, FPSGS
Vaccines prevent disease from happening or may attenuate the effects of the illness when the individual gets infected. Since the approval of vaccines for emergency use, there has been a lot of queries on its safety and use for cancer patients. Is it really safe to receive the COVID-19 vaccine even if you have cancer? Bottom line is, YES. Available data shows that vaccines are safe and fairly effective for patients with malignancy and even those receiving active treatment such as surgery, targeted therapy, chemotherapy and radiotherapy.
KEYPOINTS
- Having cancer is associated with worse outcomes when infected with COVID-19
- COVID-19 vaccines approved by the FDA are generally safe and effective for cancer patients
- Majority of patients with cancer may be vaccinated with the COVID-19 vaccine once it is available
- Despite vaccination, immunity is not absolute, therefore minimum health protocols should still be practiced
WHY VACCINATE?
Studies have shown that patients with cancer may be associated with worse outcomes when infected with the SARS COV2 virus.
In a meta-analysis done by Giannakoulis,et al, involving 46,499 patients (1,776 patients with cancer) with COVID-19 from Asia, Europe, and the United States, it reported a higher likelihood of ICU admission and higher mortality for patients having cancer as compared to those without malignancy.1
A similar study done by Saini, et. al., involving a total of 18,650 patients with both COVID-19 and cancer, showed that patients with cancer who develop COVID-19 infection have a high probability of mortality. The study concluded that appropriate and aggressive preventive measures must be taken to reduce the risk of COVID-19.2
Besides patients with cancer, close contacts and caregivers attending to this high-risk group should also be vaccinated.
WHICH VACCINE?
As of this writing, there are seven COVID-19 vaccines approved for emergency use authorization (EUA) in the Philippines. This include the mRNA vaccines - Pfizer-BioNTech COVID-19 Vaccine (Cominarty) and COVID 19 mRNA Vaccine [nucleoside modified] (Moderna); Vaccines using adenovirus vector namely ChAdOx1-S ( AstraZeneca), Ad26.COV2-S (Janssen) and Gam-COVID-Vac (Sputnik V); and the inactivated virus vaccines Whole Virion, Inactivated Corona Virus Vaccine (Covaxin) and SARS-CoV-2 Vaccine (Vero Cell), Inactivated (Coronavac). All of which show relative effectiveness against the virus.
Most of the available literature on COVID-19 vaccines and its effectiveness in cancer patients is limited to the mRNA vaccines and those with the adenovirus vectors. Generally, all the above-mentioned vaccines may be given to cancer patients as long as there is no contraindication to its components.
One publication by Thakkar, et al showed a high seroconversion rate (94%) in 200 patients with cancer after receiving a full dose of the vaccine. However, comparing to solid tumors (98%), a significantly lower rate of seroconversion was observed in patients with hematological malignancies (85%), particularly recipients following highly immunosuppressive therapies such as anti-CD20 therapies and stem cell transplantation. Moreover, a relatively lower IgG titers were observed following vaccination with the adenoviral compared to mRNA-based vaccines.3
BEST TIME TO BE VACCINATED?
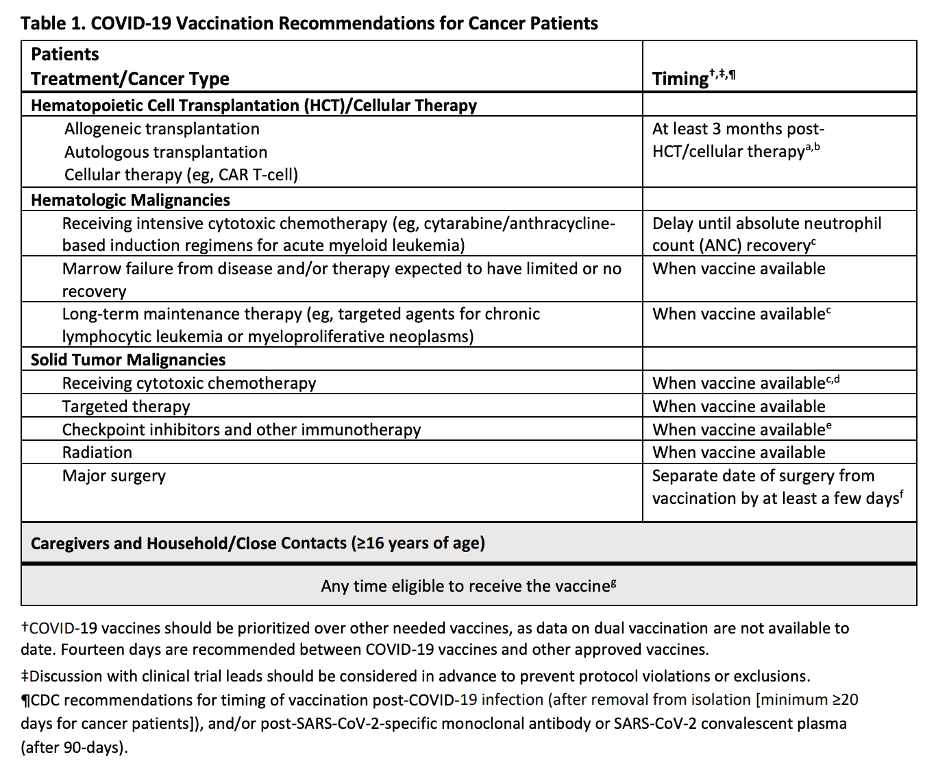
Generally, vaccines may be administered to cancer patients at any time as soon as it is available. However, there are a few exceptions such as those who are undergoing hematopoietic cell transplantation and intensive cytotoxic chemotherapy for hematologic malignancies. Those undergoing elective surgery may receive the COVID- 19 vaccine but should be delayed for a few days (5-7 days) so as not to spawn confusion whether it is a side effect of the vaccine or a complication of the surgery. If you are undergoing active treatment or scheduled for elective surgery, it would still be best to talk to your doctor regarding the timing of your vaccination.
News article: NCCN Updates on COVID 19 Vaccination: (https://www.nccn.org/guidelines/guidelines-detail?category=3&id=1457)
SIDE EFFECTS AND CONTRAINDICATIONS
Common side effects of COVID-19 vaccines are relatively mild which include pain and discomfort at the injection site, fever and chills, headache and dizziness, increase in blood pressure and rash.
The only contraindication to COVID-19 vaccination is having allergic reactions to the COVID-19 vaccines or their components.
For more information, the Department of health DOH keeps an updated list of reports for suspected adverse reactions. (https://www.fda.gov.ph/list-of-fda-issued-emergency-use-authorization/)
References
1) Giannakoulis et al., Effect of Cancer on Clinical Outcomes of Patients With COVID-19: A Meta-Analysis of Patient Data, DOI: 10.1200/GO.20.00225 JCO Global Oncology no. 6 (2020) 799-808. Published online June 8, 2020. 2) Saini et. al., Mortality in patients with cancer and coronavirus disease 2019: A systematic review, DOI: 10.1016/j.ejca.2020.08.011 3) Thakkar et al., Seroconversion rates following COVID-19 vaccination among patients with cancer, Cancer Cell(2021), https://doi. org/10.1016/j.ccell.2021.06.002 4) https://www.nccn.org/covid-19 5) https://www.fda.gov.ph/list-of-fda-issued-emergency-use-authorization

